
A metallic bond occurs when two metal atoms bond. Most chemical bonds occur as atoms fight over electrons. duntaro/iStock/Getty Images Plus Metals can even form a special bond Cesium (Cs) is the most metallic of the naturally occurring elements. Elements become more metallic as you move from right to left and also as you move from top to bottom on the periodic table. Their malleability (bendiness), ductility (stretchiness) and conductivity come from the tendency of these elements to lose electrons. In many ways, however, this weakness turns out to be metals’ strength. Metals do desire electrons, but only weakly. 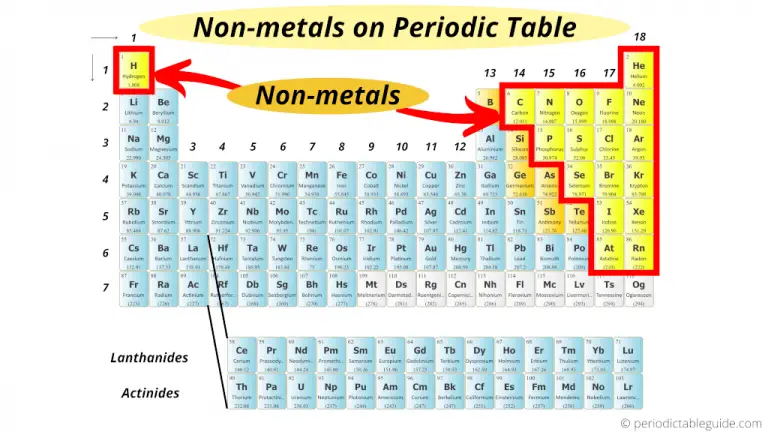 
When it comes to electron-stealing, fluorine’s a downright bully. When it comes to electron thieves, nonmetals are the best. Among these nonmetals are carbon, oxygen and nitrogen. Nonmetallic elements, therefore, have a low metallic character. This tendency to lose electrons is described as their “metallic character.” Instead of capturing a neighbor’s electrons, they usually give up their own. One might say that metals are bad thieves. Metal atoms differ from nonmetal ones in how well they steal valence electrons from other atoms. They are the outer, orbiting electrons that can become part of chemical bonds. These are called valence (VAY-lents) electrons. Different elements have different numbers of electrons that - at least in theory - can be stolen by a neighbor. It won’t steal all of them, but just enough to be stable. If it’s able, an atom will steal electrons from a neighboring atom. A thin layer of gold covers the beryllium to improve the mirrors’ reflectivity. NASA chose beryllium due to its light weight and resistance to temperature-related shape change. Its 16 hexagon mirrors, arranged like a honeycomb, are made from beryllium, a rare metal. NASA’s James Webb Space Telescope is one example of a special project the uses a rare metal. But beryllium remains more stable than most metals during such sharp temperature changes. Since telescopes work by reflecting light, any tiny change in their shape could ruin a telescope’s images. When rocketed from Earth’s relatively lukewarm air to the cryogenic (super-cold) temperatures of space, metals contract and bend. Beryllium also holds its shape in frigid temperatures. For this eye in the sky, NASA chose the rare metal beryllium (Beh-RIL-ee-um) to make its honeycomb of gold-plated mirrors. After 25 years in development, the telescope launched on Christmas morning in 2021. Sometimes, a special project - such as the $10-billion James Webb Space Telescope - may call for a fairly rare metal. But if we wanted pure sodium, we’d need a way to separate it from one of the chemicals to which it had bonded. For example, silver can be mined as pure silver. They often occur naturally in forms easy to work with. And that highlights another reason low-reactivity metals are so useful. Sodium chloride, or table salt, is a common example. Instead, we find it after it has already bonded chemically with one or more other elements. Good thing, then, that pure sodium never occurs naturally. Watch as the ingredients of table salt - sodium (a metal) and chlorine - react chemically. But pure sodium, also a metal, is so reactive it explodes on contact with water! Pure silver is so safe that we use it for jewelry and flatware. Low-reactivity metals are safer to handle than high-reactivity ones. Reactivity refers to how easily a substance reacts chemically with other substances. 
(Although most known elements are metals, they are fairly uncommon in nature.)įamiliar metals also are less reactive than most other metals. For one thing, they’re easier to find than other metals. Although more than three-fourths of the 118 elements on the periodic table are metals, we craft tools from only a few of these. That’s because familiar metals, such as iron and silver, are special. Being malleable (MAAL-ee-ah-bul), metals can be hammered into sheets without shattering. Because they’re ductile (DUK-tul), they can be easily pulled and stretched into wires without snapping. Metals also can conduct electricity.īut not all metals are equally prized. A towering skyscraper bends under a strong gust of wind, but it doesn’t snap.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
